Thermal diffusivity is a value that characterizes the rate of heat diffusion through a material. It is of great importance in the field of thermodynamics and its value is key in material selection for heat exchange applications. In this article, you will learn the thermal diffusivity meaning, its value in various materials, and compare the concepts of thermal diffusivity vs thermal conductivity.
The Meaning of Thermal Diffusivity
Thermal diffusivity indicates how quickly heat spreads within the borders of a material. It also shows the sensitivity of a material to temperature changes. Because it is a measure of the heat transfer rate, it closely relates to another material feature, which is thermal conductivity (k). In addition, it is also a function of the material’s density (ρ) and specific heat capacity (cp). From Physics, it is common knowledge that higher densities mean lower diffusion rates, due to the tight packing of atoms/molecules. Thus, dense substances have low diffusivity. On the other hand, specific heat capacity represents how much heat an atom/molecule can hold per time. The higher this value, the less likely it is for the material to transmit heat, thus, a lower diffusivity value. Mathematically, thermal diffusivity (α) is expressed as the ratio of heat conducted to heat stored.
![]()
Diffusivity in Various Materials
Thermal diffusivity is important in industries when selecting materials to deliver desirable heat transfer properties. A heat sink, which is a material with high diffusivity, serves to quickly take heat away or into a system, without storing much heat. Whereas insulators, which have low diffusivity values, ensure heat loss from a body is minimal. Below are some diffusivity values for common materials that play a vital role in a variety of applications.
| Material | Thermal Diffusivity (mm2/s) |
| Pyrolytic Graphite (parallel to layers) | 1,220 |
| Helium (300 K, 1 atm) | 190 |
| Silver (99.9%) | 165.63 |
| Gold | 127 |
| Copper (25 °C) | 111 |
| Aluminum | 97 |
| Water vapor (400 K, 1 atm) | 23.38 |
| Air (300 K) | 19 |
| Steel (1% carbon) | 18.8 |
| Steel (Stainless 304A at 27 °C) | 4.2 |
| Pyrolytic Graphite (normal to layers) | 3.6 |
| Glass (window) | 0.34 |
| Water (25 °C) | 0.143 |
| PTFE | 0.124 |
| Rubber | 0.089 – 0.13 |
| Wood (yellow pine) | 0.082 |
| Engine oil (saturated liquid at 100 °C) | 0.0738 |
| Alcohol | 0.07 |
From the standard values on the table, there are few key takeaways. The value for a material varies according to the direction of heat transfer. For example, pyrolytic graphite is a good transmitter of heat when it is travelling parallel to the graphite layers. But when traveling perpendicular to the graphite layers, heat transfer is poor. Also, when the microstructure and temperature of materials change, such as in steel and water respectively, the diffusivity values alter.
Methods of Determining Thermal Diffusivity of a Material
The measurement techniques for determining thermal diffusivity of materials are easier to carry out relative to those for measuring thermal conductivity. As a result, thermal diffussivity measurements are predominant and provide a means to estimate both diffusivity and conductivity. Some of these techniques include the laser flash method, thermal wave interferometry, and infrared thermography.
Laser Flash Method
This is an effective approach to determining thermal diffusivity for different materials. It involves hitting a plane-parallel sample with a short burst of energy on one side, while monitoring the temperature change from the other side. Other parameters monitored during this method are the time and thickness of the sample. Below is an ideal setup for this method, with an infrared (IR) detector, which helps provide temperature measurements from the sample.
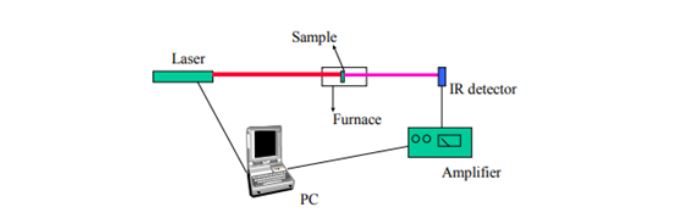
Although this method is widely applicable, it has the limitation of requiring a sample of specific geometry.
Thermal Wave Interferometry
Thermal wave interferometry is ideal for thermal diffusivity measurements of coatings and thin slabs. Moreover, it involves using a modulated laser to heat a two-layer sample that has a surface with a coating and another without. Due to the thermal mismatch of the surfaces, there will be reflection and transmission of the wave in varying manners. This varying temperature distribution is captured by an infrared (IR) detector and analyzed to produce information on both the thickness and diffusivity of the sample.
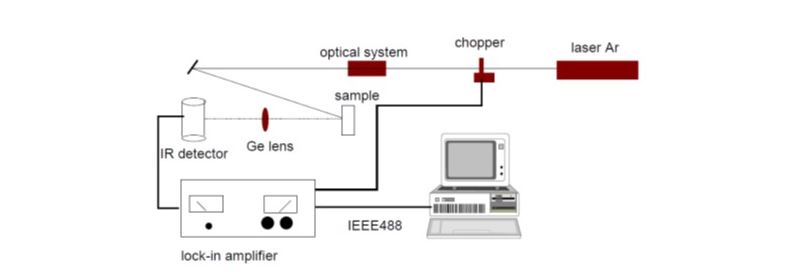
Infrared Thermography
This method entails using an instantaneous circular Gaussian source to heat a plate-like sample from one end. Then, analysis occurs on the spatial distribution or phase image of the temperature at either the front or rear surface of the sample.
Moreover, there are three approaches to infrared thermography, which are the single-flash method, lateral thermal waves, and spatially resolved diffusivity measurement. Of these three, the lateral thermal waves and spatially resolved measurements record the most accurate values with an error of ± 0.0006×10-4 cm2/s.
Thermal Diffusivity vs Thermal Conductivity
Thermal diffusivity and thermal conductivity are common concepts within the scientific community for quantifying heat transfer measurements. They both quantify how easy it is for heat to transmit via a material. However, there are some differences between them, which the table below highlights.
| Thermal Diffusivity | Thermal Conductivity |
| It shows how rapidly heat will flow within a material. | Indicates how much heat will flow through a material. |
| Its unit of measurement is m2/s. | The units for conductivity are W/mK. |
| Thermal diffusivity accounts for a material’s ability to store thermal energy. Thus, it helps to determine when the material will attain steady-state temperature distribution. | Conductivity accounts for the overall amount of heat transfer, without differentiating the stored component. So, it is not useful in evaluating steady-state and transient temperature distribution. |
| Mathematically, diffusivity is denoted by α. | Mathematically, k denotes conductivity. |