The specific heat of ice measures the amount of heat needed to raise the temperature of a particular substance in a particular form by one degree Celsius. This article provides the specific heat of ice in standard units and answers common questions about the specific heat.

What is the specific heat of ice in joules?
Since heat measures in Joules (J), mass in grams (g), and temperature in degrees Celsius (°C), we can deduce that specific heat measures in Joules per g times °C.
For standard pressure and temperature (1 atm, 32 oF), ice has a specific heat capacity of around 2.03 joules per gram of Celsius.
What is the specific heat of ice in calories?
Ice has a specific heat of 0.5 g/°C in calories. Because ice has a specific heat of 0.50 cal/g-°C, it takes 0.50 calories to raise 1g of ice to 1°C. To increase the melting point of 50g by 1°C, it would take 50 x 0.50 calories and 10 × 50 x 0.50 = 250 calories.
What is the specific heat of ice in kJ/kg∙°C?
The specific heat capacity of ice is 2.10 kJ/kg°C, which is the amount of heat required to increase the temperature of any particular substance in a particular form by one degree Celsius.
What is the specific heat of ice in J/kg∙°C?
The specific heat of ice is 2093 in Joule per kilogram per degree Celsius.
What is the specific heat of ice vs. water?
Water has a specific heat capacity of 4.187 kJ/kg K. In contrast, ice has a total heat of 2.108 kJ/kg K. Thus it takes 2.108kJ to raise the temperature of 1 kg of ice by 1K.
Why Does Ice Have a Lower Heat Capacity Than Liquid Water?
Ice has a lower heat capacity vs. water. This is true because as a liquid, water has many directions to flow and absorb heat. Thus, more overall surface area must be heated to raise the overall temperature.
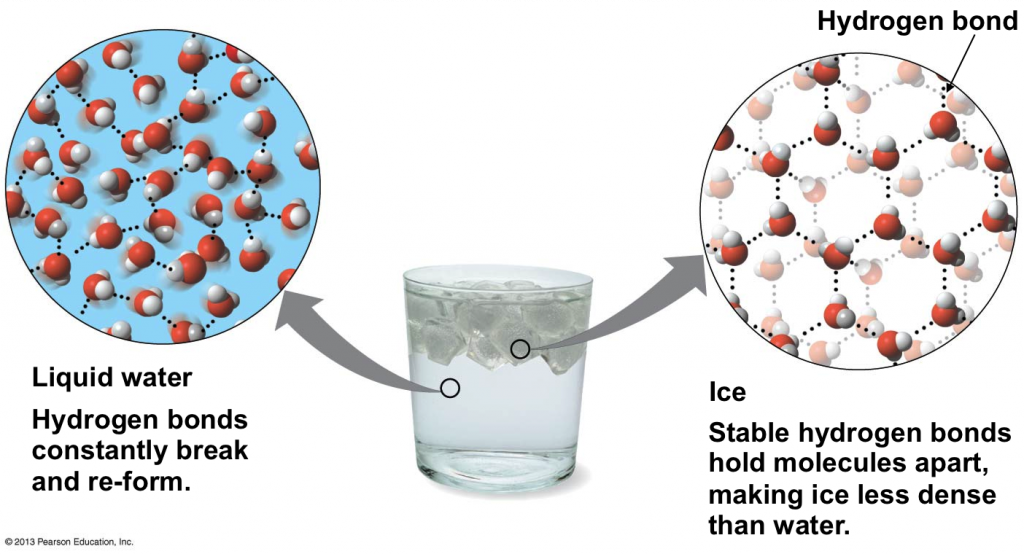
The surface area of ice, on the other hand, does not alter due to its more rigid structure. When ice heats up, the heat energy has to go somewhere, breaking down the solid’s shape and melting it into water.
To conclude, the energy given to water in liquid form distributes between different forms of kinetic energy, while that given to ice only transforms into one form of kinetic energy. Therefore, more power is needed to raise the general temperature of the water.
Ice’s Thermal Properties
Thermal characteristics are those of a substance related to its heat conductivity. Another way, when heat is introduced to a material, it exhibits specific characteristics. Thermodynamic properties are a subset of material physical properties.
Ice is simply water frozen into a solid state. It might seem transparent or opaque bluish-white depending on contaminants such as soil particles or air bubbles. Here are the ice thermal properties:
Melting Point
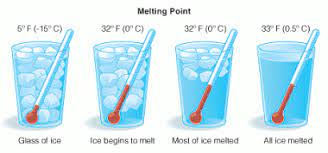
Ice has a melting point of 0°C or 32°F. It’s worth noting that this measurement corresponds to the typical atmospheric pressure. Melting is defined as the transition of a substance from a solid to a liquid state. The melting point, in a sense, is the temperature at which this phase change occurs. The melting point also defines a condition where the solid and liquid can exist in equilibrium. It is difficult to determine the melting point for various chemical compounds and alloys since they are usually a mixture of different chemical elements.
Thermal Conductivity
The thermal conductivity of ice is 2.22 W/(m·K). The thermal conductivity, k (or λ), is a property that measures the heat transport characteristics of solid materials in W/m.K. It assesses a substance’s ability to transfer heat by conduction through a medium. Fourier’s law holds true for all matter, regardless of its state (solid, liquid, or gas). As a result, it also applies to liquids and gases.
Specific Heat
In addition to the previously specified readings, the specific heat of ice registers at 2040 J/g K. Specific heat, often known as specific heat capacity, is a thermodynamic parameter connected to internal energy. The intense properties cv and cp are partial derivatives of the internal energy u(T, v) and enthalpy h for pure, simple compressible substances (T, p).




